
Reactions occur in the human body between various antigens and antibodies. The presence of heavy metals can beĮliminated by precipitating them as their hydroxides and sulfides by using In addition to chrome yellow (PbCrO4), examples include Prussian blue (Fe7(CN)18), the reddish-orange color vermilion (HgS), and green color veridian (Cr2O3). Reactions are used in treating wastewater. Reactions have a great application in the extraction of elements like magnesium In this experiment, you will monitor conductivity during the reaction between sulfuric acid, H 2 SO 4, and barium hydroxide, Ba(OH) 2, in order to determine the equivalence point.From this information, you can find the concentration of the Ba(OH) 2 solution. Green precipitate indicates the presence of Fe2+ and Cr3+.Ĭoncept of precipitation is also used in titrations and gravimetric analysis. 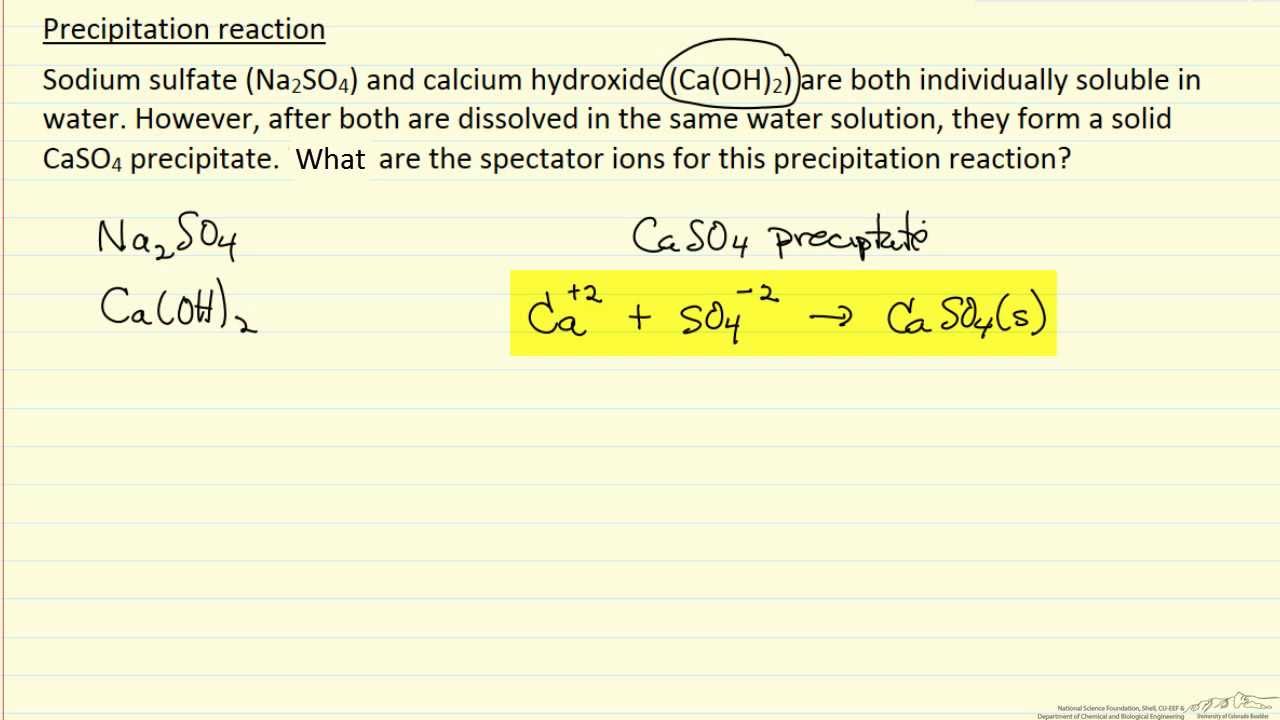
Gelatinous precipitate indicates the presence of Al3+ ions, and Ions, are precipitated by using ammonium hydroxide and ammonium chloride.įormation of red precipitate indicates the presence of Fe3+, white The detection of many cations andĪnions is done by precipitating them in a solution. Reactions are used in qualitative analysis. Organic synthesis, various precipitation techniques are used to isolate and Reactions are used for preparing and isolating many useful compounds. Thus, it can be concluded that the net ionic equation of a precipitation reaction shows the precipitate formed as a product from its corresponding cations and anions. 
The general form of double displacement reaction showing precipitation reaction is:Ī B ( a q ) + C D ( a q ) → A D ( s ) + C B ( a q ) AB_ A ( a q ) + + D ( a q ) − → A D ( s )
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
